The heavier atomic weight of tritium has an effect on the physical properties of this hydrogen isotope. For example, tritium has a boiling point of 25K (-415°F; -248°C), compared with ordinary hydrogen's boiling point of 20.4K (-423°F; -252.8°C). Molecules containing tritium show similar variances. Tritium Protium Ordinary hydrogen is knows as protium. It has one electron,one proton but it has no neutron. Mass number: 1; Charge number: 1; Symbol: 1 H 1 Percentage in natural hydrogen 99.98% Structure Deuterium Heavy hydrogen is known as deuterium.
Tritium
HTritium (H-3) has an atomic number of 1 and a half-life of 12.7 years. It is a low energy 0.019 Mev (max) beta emitter. The emitted beta radiation cannot penetrate the outer layer of dead skin. There is no external exposure hazard and no required shielding. Tritium has an atomic number of 1, like other hydrogen isotopes, but it has a mass of about 3 (3.016). Tritium decays via beta particle emission, with a half-life of 12.3 years. The beta decay releases 18 keV of energy, where tritium decays into helium-3 and a beta particle. As the neutron changes into a proton, the hydrogen changes into helium.
3
Properties
General Info:
Radioactive Decay:
Specific Activity:
Half-Life:
Tritium is an isotope of Hydrogen. Most hydrogen is made up of one proton, and an orbital electron, but tritium has two extra neutrons in the nucleus.
The radioactive decay product of tritium is a low energy beta particle, which is similar to an electron. This reduces the energy in the nucleus and the atom, now a helium atom (3He), is left more stable.
9,650 curies (3.57×1014 Bq) per gram.
12.32 years
Tritium Sources
Environmental:
Production:
Cosmic rays interact with nitrogen or with deuterium and form tritium and carbon. These are primarily interactions that happen in the upper atmosphere and the tritium falls to earth as rain.
Tritium is produced in nuclear reactors by neutron activation of lithium-6. Tritium is also produced in heavy water-moderated reactors whenever a deuterium nucleus captures a neutron.
Exposure
Tritium has a single electron the same as the more abundant forms of hydrogen. This causes tritium to react chemically to form compounds in the same manner as hydrogen. The two primary forms that personnel will likely to be exposed to are HT (which is similar to hydrogen gas) or HTO (tritiated or heavy water). Of these two forms, the HTO is the only form that is a significant exposure hazard. HT gas is inhaled and exhaled with only of 0.005% of the activity being deposited in the lungs.
The uptake of HTO vapor is near 100% for inhalation and ingestion. Tritium can also enter the body by absorption through the skin or open wounds. Skin contact should always be minimized to prevent absorption. Tritium will also be absorbed into materials such as gloves, clothing and metal. If not properly controlled, these contaminated materials can present an additional exposure source by releasing tritium when in contact with skin. On an up-take, some of the tritium can be held as organicly bound material, but the dose from this bound tritium is much less than the free tritium.
HTO is in the form of water, so one to two hours after an uptake, it will be evenly distributed through out the body's fluids. The amount of time it takes for half of the activity to be physically removed form the body is the biological half life. The biological half life of tritium varies significantly because of variations in bodily excretion rates, temperature dependence and fluid intake. Biological half-life of tritium is about 9.4 days, often rounded to 10 days. This can be shortened to 2-3 days (Fig 1) with ten fold increase of liquid intake (2 liters to 20 liters), or in serve cases to 4-8 hours by using dialysis machines.
Source: http://www.physics.isu.edu/radinf/tritium.htm
Improper Disposal of Tritium
The Department of Environmental Protection (DEP) recently completed a comprehensive two-year study at 54 landfills within the Commonwealth, testing for the presence of radioactive materials in landfill leachate. The study was performed as a follow-up to DEP's new requirements for radiation monitoring at all solid waste management facilities in Pennsylvania. Although sample results quantified certain naturally occurring elements within natural background levels, including uranium, thorium and potassium, above-normal levels of tritium were noted in leachate at many facilities.
The source of higher-than-background levels found in landfill leachate samples was presumed to originate from the improper disposal of self-luminescent exit signs found in construction/demolition (C/D) waste and other solid waste streams. There are no other known sources of tritium in industrial or consumer products that would cause elevated levels of tritium in landfill leachate. Thus, it is apparent that tritium exit signs, which when new may contain up to 25 curies, or 25,000,000,000,000 picocuries (pCi) of tritium, are entering landfills via municipal or residual waste streams. A single tritium emergency exit sign has the potential to cause the tritium levels observed.
Source: http://www.dep.state.pa.us/brp/radiation_control_division/Tritium-LF-Leachate-Rpt_final_SECEREF_3-31-2010.pdf
Also found in: Thesaurus, Medical, Financial, Encyclopedia, Wikipedia.
trit·i·um
(trĭt′ē-əm, trĭsh′ē-)n.tritium
(ˈtrɪtɪəm) n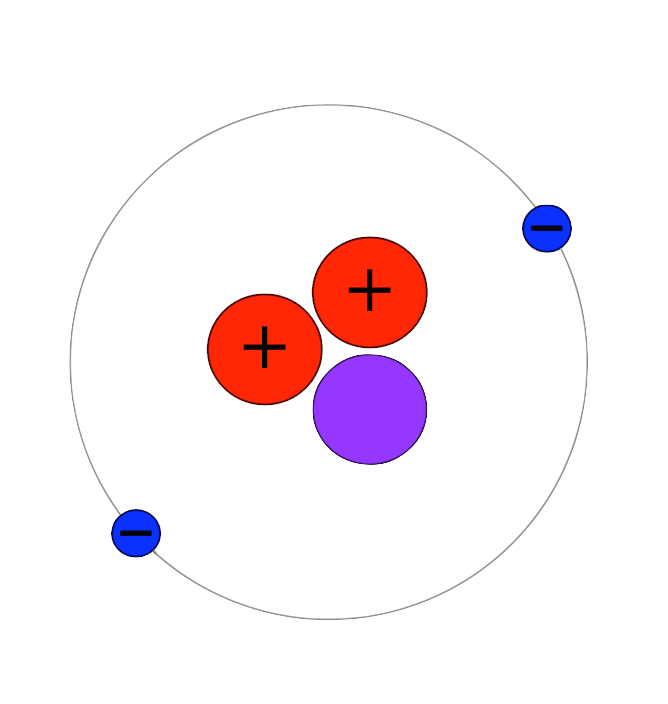
trit•i•um
(ˈtrɪt i əm, ˈtrɪʃ-, ˈtrɪʃ əm)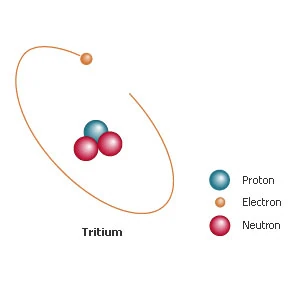 n.
n. trit·i·um
(trĭt′ē-əm, trĭsh′ē-əm)tritium
| Noun | 1. | tritium - a radioactive isotope of hydrogen; atoms of tritium have three times the mass of ordinary hydrogen atoms atomic number 1, H, hydrogen - a nonmetallic univalent element that is normally a colorless and odorless highly flammable diatomic gas; the simplest and lightest and most abundant element in the universe |
Tritium Atomic Number Mass
tritium
[ˈtrɪtɪəm]N → tritiomtritium
Want to thank TFD for its existence? Tell a friend about us, add a link to this page, or visit the webmaster's page for free fun content.
Link to this page:

Tritium Periodic Table
